Professor of Harbin Institute of Technology jointly developed a new type of super resolution structured light microscope technology, which is suitable for living cell imaging.
On November 16, the latest paper participated in by Haigui Professor Li Haoyu of the Institute of Instrument of Harbin Institute of Technology was finally published on Nature Biotechnology. "As long as we are curious about the world, we will never stop pursuing resolution," he said.
The paper is entitled "Sparse deconvolution improves the resolution of living cell super resolution fluorescence microscope". Professor Li Haoyu of Harbin Institute of Technology and Professor Chen Liangyi of Peking University are the co corresponding authors of the paper.
The publication of this paper also means that Mr. Li Haoyu and his collaborators have made breakthrough in the field of optical super-resolution microscopic imaging technology. Under the condition of low phototoxicity, the resolution of the structured light microscope was increased from 110 nm to 60 nm to achieve ultra fast, long-term super resolution imaging of living cells.
Li Haoyu said that optical superresolution broke the physical diffraction limit of light. The 2014 Nobel Prize in Chemistry was awarded to scientists who solved the above physical limits by manipulating fluorescent proteins.
Up to now, the physical and chemical methods to achieve super-resolution imaging have reached the bottleneck, and the pursuit of higher microscope resolution is facing great challenges. This research is actually a generalized mathematical algorithm framework, which can double the imaging resolution of several commonly used fluorescence microscope instruments.
Take the turntable confocal microscope as an example. The normal resolution of its commercial instrument is about 160 nm. With this achievement, it can be improved to about 80 nm. Therefore, the team has pushed forward the bottleneck limit resolution that can be reached before from the perspective of optimization calculation.
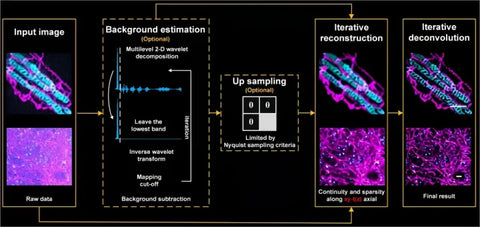
In the research, he and his team proposed a computational microscopic imaging method, which can break through the optical diffraction limit. The basic principle is that with the help of the forward physical model of fluorescence imaging and the compression perception theory, when using the double constraints of sparsity and space-time continuity, a general image solution method can be established. The framework is equipped with sparse deconvolution technology, which breaks through the hardware limitations of the previous optical super-resolution microscopic system, Spatial and temporal resolution and spectrum can also be expanded.
Based on this technology, Li Haoyu and others, together with Chen Liangyi's team from Peking University, have jointly developed an ultra fast structured light super-resolution fluorescence microscope system (Sparke SIM), which is characterized by super-resolution, high-throughput, low toxicity, non-invasive, etc. When in high-speed imaging, the system can not only achieve a resolution of 60 nanometers, but also achieve ultra long time dynamic and fast imaging of living cells for more than one hour.
With the help of the imaging technology in this achievement, the dynamic network of actin, the fast behavior of lysosomes and lipid droplets in deep cells can also be observed, and the fine movement between the inner and outer membranes of two-color mitochondria can also be observed.
To sum up, on the basis of physical and chemical methods, Li Haoyu and his team first proposed a general algorithm model to break through the optical diffraction limit from another point of view of mathematical calculation. This is not only an original research from 0 to 1, but also has developed a super resolution microscope instrument with the highest resolution, longest imaging time and fastest imaging speed in living cell optical microscopy.
Surprisingly, this technical framework has also been proved to be applicable to most current fluorescence microscope imaging system modes, which can achieve nearly twice the stable spatial resolution improvement, providing a new generation of biomedical super-resolution imaging instrument for precision medicine and new drug research and development, making it possible to significantly accelerate the high-precision characterization of disease models in the future.

Share:
Q&A of Storm2 Power Bank
Understanding Structured Light and 3D Scanning